This is the reflection for the second week (January 20-26) since I am a bit behind (ha!). The second weeks lesson was really fascinating because it was covering primarily paleo-climate information. Prior to week 2 readings I had only the haziest notion of paleo-climate, so it was really informative.
I understand that of necessity the paleo-climate models they provided us were simplified (nothing about ocean currents for example in discussing mechanisms of climate), but despite being simplified they were quite powerful in providing explanations for the major long term changes in earth's climate.
First important discovery: our sun, Sol, puts forth substantially more energy today than it did 4.4 Billion years ago when the solar system and Earth was formed. Now before someone goes "ah ha! that's what's driving global warming today" let's quickly put that to rest. We are talking a slow, gradual warming for the entire 4.4 Billion year period - which covers both much warmer and much colder geological periods, and has little to do with the dramatic increase in temperatures observed in just the past 150 years since the start of the industrial age.
So 4.2 Billion years ago (once the surface of the planet settled down enough to result in rock evidence we can still find today), the sun was substantially weaker, and earth received substantially less solar radiation. The estimate is 20 to 30 percent less energy output 4.2 Billion years ago (https://www.sciencenews.org/article/faint-young-sun). This creates a bit of a puzzle: if we were to suddenly lose 20 to 30 percent of the sun's energy today, our world would plunge into a bitter freeze. The earth's surface would be so cold that there would be no liquid water left. But geologists and paleo-biologists can demonstrate to us in the form of fossils, and evidence of rain and water weathering that there was abundant liquid water on the surface of the earth 4.2 Billion years ago. So how come the earth was warm enough for liquid oceans, rain, streams and lakes even though there was less radiation from the sun. The most accepted hypothesis is that the earth's atmosphere was much denser and had much higher levels of carbon dioxide, methane, and combinations of nitrogen and oxygen that facilitated heat absorption. One source of these greenhouse gases would have been volcanoes. This is still an area of research and discussion to find the precise mechanisms for the warming.
A new puzzle arises when one asks the question, well if the earth were as least as warm as it is today, with less sunlight and more greenhouse gases, why didn't the earth get consistently hotter over time? The key to that is in chemical and geological processes that remove carbon dioxide from the air and lock it up in rocks in the earth's surface. Particles of carbon dioxide are absorbed by water vapor and create rain, a lightly acidic rain (carbonic acid rain) that over time weathers (chemically wears down) the rocks of the earth's surface. Rain run-off in streams and rivers carries that rock and carbon bearing water into the oceans where the carbon and other minerals from the rock are used by tiny living creatures in the oceans to build their bodies and their shells. When they die, their remains filter to the bottom of the ocean and provide the sediments that become sedimentary rocks. Constant weathering and the growth of living species removes carbon from the atmosphere over the billions of years that the sun grew brighter.
About 2.2 Billion years ago a new puzzle emerges. For the first 2 billion years the earth's climate experienced swings from warmer to cooler, with the shrinking and growing of the earth's icy poles bearing witness to those swings. But at about 2.2 Billion years ago the geological evidence strongly supports the idea that the whole surface of the earth froze over, creating what is called "snowball earth". There are a variety of things that might have caused a cooling cycle, and physicists have determined that if as much as two-thirds of the earth's surface became covered with ice and snow, then the dramatically increased albedo (reflection back into space of sunlight) would reach a tipping point and there would be nothing to stop the earth from freezing entirely, which is apparently exactly what happened. The true puzzle becomes, how did the earth emerge from that frozen state to return to millions of years of much warmer climates? The key seems to be in the continuation of volcanic action throwing more greenhouse gasses into the atmosphere, but with all surface water frozen there was no rain to wash the carbon dioxide out of the air, create carbonic acid rain, and engage in rock weathering that would sequester the carbon in the earth's surface. So the concentrations of greenhouse gases increased, and increased and increased until the air was warm enough to begin melting the ice ball.
To me the most significant aspect of all this information is that the basic mechanism of climate - sunlight and greenhouse gases were the same 4.2 billion years ago as they are today, the only difference is that today we industrial humans are inputting significant additional carbon, methane, etc. into the atmosphere with our economic activity. We are taking carbon that was sequestered in the earth's crust for billions of years, hauling it out, burning it and returning it to the atmosphere. So that in 2013 the Mauna Loa monitoring station measured 400 ppm of atmospheric carbon dioxide, a level not seen on earth for the past 2 million years.
Sunday, February 2, 2014
CLIMATE CHANGE - UNIVERSITY OF EXETER - Week 2
Saturday, January 18, 2014
CLIMATE CHANGE - UNIVERSITY OF EXETER - Week 1
Even though I've been teaching about climate change for 15 years, there's still a lot of the science I don't fully understand, and I'm also always looking for ideas on how best to communicate complicated ideas about the environment to my undergraduate students, and how to engage them more fully in learning. So I happened across this link to Future Learn an on-line learning consortium of British universities, and in particular to an undergraduate oriented course on climate change. It's eight weeks long, free, and so far seems quite engaging. This is not my first on-line learning experience (I also teach mostly on-line classes) but it is my first experience with a MOOC, and with a course that has such high production values.
One of the things that we are encouraged to do in this course is to use a blog to engage in "reflective learning." At first I thought about creating a whole new blog, but that seemed redundant given that Blue Island Almanack was just sitting here unused for the past four years. So here I am!
Week 1 covered a lot of basic material. So I was surprised to find that there were a number of things that were new to me, or that I understood better by the end of the week than previously because of the skill with which they had been explained.
The first little surprise was the explanation for why "greenhouse" is not the best analogy for how our atmosphere holds heat. The glass of a literal greenhouse does not prevent long wave (heat) radiation from leaving the greenhouse. This was new to me, simplistic explanations given to me years ago said that the glass prevented the heat from escaping, turns out that is not quite correct. Long wave (heat) radiation does escape through greenhouse single pane glass. However, the glass does provide a physical barrier to wind that would remove heat by convection. This makes so much sense to me - I spent two summers of my life (1970 and 1971) working in greenhouses planting and taking cuttings from chrysanthemum plants, and know what the heat of a greenhouse is like.
A better analogy for how the earth's atmosphere retains heat is NASA's temperature regulating blankets . This high tech blanket is embedded "millions of invisible microcapsules that absorb excess heat when you are hot and release the stored heat when you are cold, ensuring a comfortable temperature and humidity." In the earth's atmosphere the "microcapsules" that absorb and release heat are molecules of the various greenhouse gases: water vapor, carbon dioxide, methane, ozone, CFC's and nitrous oxide. Which brings up another new factor I encountered this week: I'd never heard water vapor called a "greenhouse" gas previously. It is different from the other greenhouse gases listed, because it changes in concentration as temperature changes. Water vapor does absorb and release heat, but water vapor increases when temperature increases and decreases when temperature decreases, so it is an important feedback greenhouse gas, but not a "forcing" gas that changes concentration due to non-climatic events.
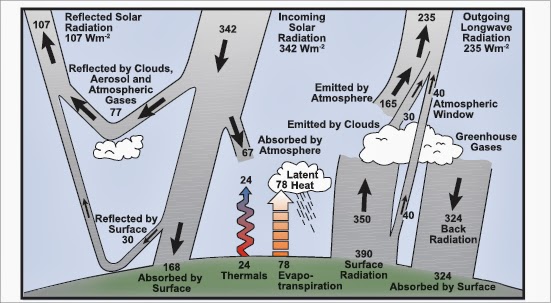
The most interesting thing I got from this week was this diagram that helped me understand several important aspects of our atmosphere and how it promotes life and affects climate.